
CITI Access
- Visit the CITI Program website and sign in.
- If you do not have a CIT account, create one using your ACU email address and choose ACU as your institutional affiliation during registration.
- Once logged in, select View Courses.
Under the “Abilene Christian University” section, click Add a Course to enroll.
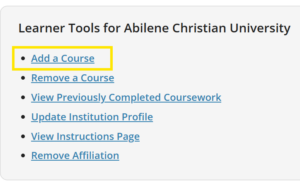
Please find the CITI guidance PDF for reference.
For assistance with training requirements, please contact: orsp@acu.edu.
CITI Curriculum Selection
Human Subjects Research (IRB)
Investigators conducting research involving human participants must complete the required Human Subjects Research (HSR) Training before submitting their research to the Institutional Review Board (IRB).
To access the training, on the Select Curriculum page, select the Social/Behavioral/Education box under Question 1- Human Subject Research (HSR). 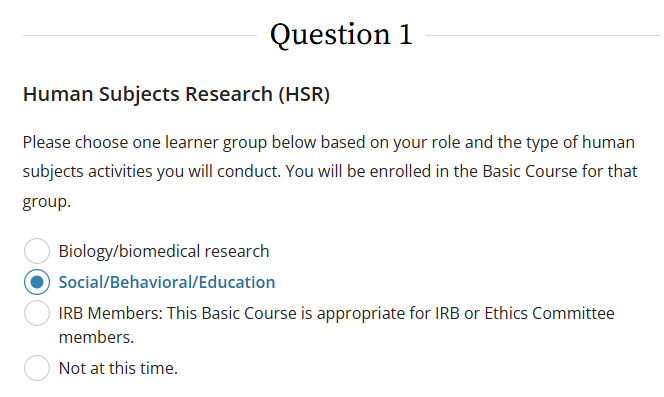
Responsible Conduct of Research (RCR) Training
Certain federally funded investigators, trainees, and students are required to complete Responsible Conduct of Research (RCR) training in accordance with Sponsor requirements (e.g., NIH, NSF). For sponsor-specific requirements and applicability, please consult the sponsor guidelines or contact ORSP.
To access the training, on the Select Curriculum page, select the applicable topic(s) under Question 2- Responsible Conduct of Research (RCR). RCR must be renewed every four years.

Animal Care and Use (IACUC)
Investigators conducting research involving live vertebrate animals must complete required animal care and use training before IACUC protocol approval. For more information, please visit the Animal Care page.
To access the training, on the Select Curriculum page, select the applicable topic(s) under Question 7- Animal Care and Use (ACU).
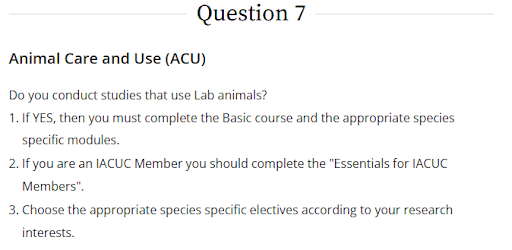
Institution Biosafety Committee (IBC)
Institutional Biosafety Committee (IBC) training typically requires all personnel working with recombinant DNA, synthetic nucleic acids, or biohazardous agents to complete initial safety training, often through CITI Program.
For more information, please contact the risk management office at risk@acu.edu.
Conflict of Interest (COI) Training
All investigators are required to complete Conflict of Interest (COI) training in accordance with institutional policy.
Note: COI training is separate from the annual COI disclosure requirement.
To access the training, on the Select Curriculum page, select the applicable topic(s) under Question 4- Conflict of Interest (COI).

Research Security Training (RST)
Certain Investigators are required to complete Research Security Training in accordance with federal regulations and institution policy.
Required Training and access: NSF Website – Research Security Training
- Go to NSF.gov/research-security/training (no login required)
- Complete all four training modules
- Submit the completion certificate to ORSP
